Our solutions
Our customers
.avif)
Executive Summary
Challenge
High-throughput media optimization requires matching throughput for experiments and analytics. While MediOP's automated suspension platform can generate hundreds of conditions in parallel, conventional, labor-intensive IgG quantification methods (requiring sample preparation and purification) quickly become the bottleneck that slow optimization cycles and limit the effectiveness of high-throughput screening.
Multus sought to build an end-to-end automated workflow where titer measurement kept pace with experimental design. The solution required a rapid, accurate, plate-compatible IgG assay that would generate decision-ready data at scale. Valita Titer assay’s fluorescence-polarization technology offered the key advantage: direct measurement from crude culture samples with no purification steps, combined with excellent correlation to established methods like Protein A HPLC and BLI. This capability would enable MediOP's machine learning algorithms to rapidly converge on optimal formulations by providing the high-quality data essential for Bayesian optimization.
Project Overview
Build and validate an automated media optimization workflow for CHO-DG44 cells producing monoclonal antibodies, targeting:
The project was structured in two phases using the MediOPsuspension platform with integrated Valita Titer assay analytics:
Phase 1: Exploration
Phase 2: Optimization
Throughout both phases, titer was quantified using the Valita Titer assay, providing the rapid analytical turnaround essential for iterative optimization.
Our Solution
Using our AI-driven MediOP™ platform integrated with Valita Titer assay technology, we developed an end-to-end automated workflow. The platform leverages Bayesian optimization algorithms to systematically explore complex ingredient design spaces, identifying formulations that simultaneously optimized for higher titer and lower cost. By enabling direct analysis of crude culture samples, the assay reduces sample handling and eliminates centrifugation-dependent preparation steps common in traditional titer methods.
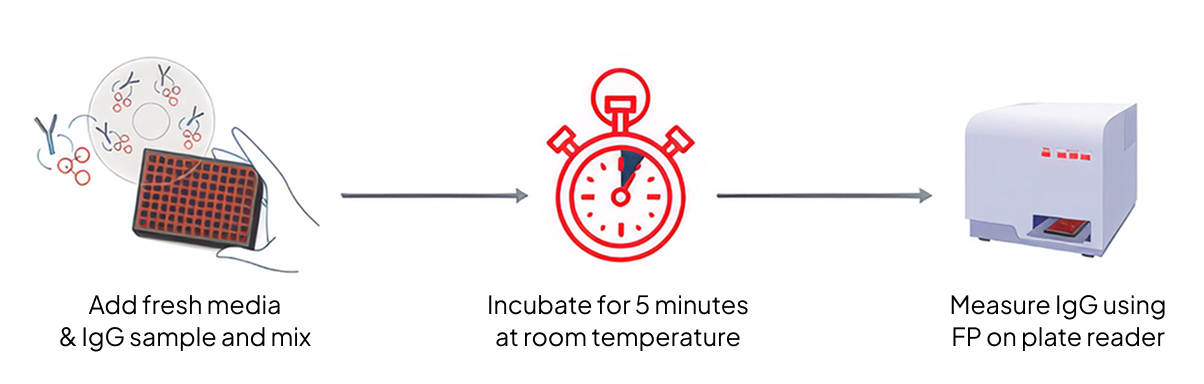
The integration of Valita Titer assay was critical to enabling true high-throughput optimization. Its simple add-mix-read protocol eliminated time-consuming sample preparation, allowing <7 minutes of sample processing per plate while its fluorescence-polarization technology provided accurate IgG quantification that correlated strongly with gold-standard methods (Protein A HPLC, BLI). The assay's compatibility with standard 96-well plate readers, integration with our in-house automation platforms and ability to measure directly from crude culture samples meant that reliable titer data could be generated at the same pace as experimental conditions, creating as eamless feedback loop for MediOP's optimization algorithms.
The Results
The MediOP platform's integration with Valita Titer assayenabled comprehensive screening and optimization across 150 formulations:


Key performance highlights include:
Future Outlook
Building on the success of integrating Valita Titer assay with the MediOP platform, Multus plans to expand this optimization framework across multiple dimensions. The platform will be extended to additional CHO cell lines, and diverse molecular formats including multispecific antibodies, fusion proteins, and other Fc-containing therapeutics to validate robustness across cell types and products. The proven compatibility between MediOP's automated workflows and Valita Titer assay’s analytics provides a strong foundation for these expanded applications.
Future optimization cycles will incorporate product quality attributes such as glycosylation patterns, charge variants, and aggregation profiles, alongside bioreactor scalability criteria, enabling more predictive transitions from microscale screening to manufacturing scale. Additionally, Multus is exploring integration of complementary high-throughput analytical methods alongside Valita Titer to create multi-parameter optimization workflows for complex media development challenges.
By combining MediOP's automated experimentation with cost-aware analytics and Valita Titer assay’s high-throughput assay capabilities, Multus provides biologics developers with a scalable route to accelerated CHO media optimization, supporting both early-stage development and commercial media design.
Technical Note: Titer quantification throughout this study utilized Valita Titer (Beckman Coulter Life Sciences) fluorescence-polarization assay for Fc-containing IgG in 96-wellformat. The assay enabled direct measurement from crude culture samples without purification, providing rapid analytical turnaround essential for high-throughput optimization. Strong correlation with Protein A HPLC and BLI methods validated data quality throughout the study.
See here for more information: ValitaTiter Assays for IgG Quantification [EN1]
The MediOP™ suspension culture platform (Multus Biotechnology) integrated automated liquid handling and Valita Titer assay analytics for CHO DG44 cells producing IgG monoclonal antibodies in fed-batch format with bolus feeding every 48 hours(cultures terminated at 70% viability).
Beckman Coulter, the stylized logo, and the Beckman Coulter product and service marks mentioned herein are trademarks or registered trademarks of Beckman Coulter, Inc. in the United States and other countries. Valita, Valita Titer and the Valita Cell logo are trademarks of Valita Cell Ltd.in the United States and other countries. Valita Cell is a Beckman Coulter Company.
Sign up for email updates: